NMN Antioxidant Activity Prevents Liver Injury in New Study
Perhaps the most distinguishing feature setting us apart from other species is our ability to adapt. This includes the ability of our cells to effectively respond to environmental and metabolic stressors by activating protective processes that maintain homeostasis. Scientists propose that this cellular adaptability declines with age and underlies the aging process.
A report published in the Journal of Proteome Research from Tsinghua University in China suggests that NMN can combat aging by restoring cellular adaptability. Luo and colleagues show that NMN protects aged mice from liver injury by reducing oxidative stress via sirtuin3 activation. Further experiments show that NMN mediates the restoration of protective adaptations that reestablish liver homeostasis in aged mice.
NMN Prevents Liver Damage by Reducing Oxidative Stress
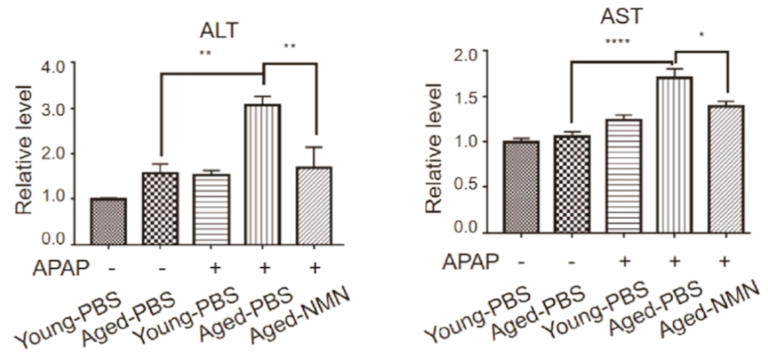
To study the effects of NMN on liver injury, Luo and colleagues injected 96-week-old mice (equivalent to about 65 human years) with 500 mg/kg of NMN. Liver injury was induced by exposing the mice to acetaminophen (APAP). Excessive amounts of APAP cause severe liver toxicity, the leading cause of liver failure. APAP led to high blood levels of ALT and AST, markers of liver damage, in the aged mice. However, NMN treatment restored ALT and AST levels back to youthful levels.
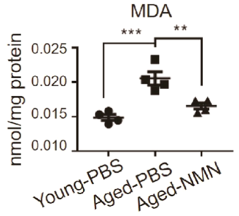
Oxidative stress from harmful reactive oxygen species accumulation causes damage to macromolecules (carbohydrates, proteins, DNA/RNA, and fats) with increasing age. When oxidative stress damages fats (lipids), the concentration of a protein called malondialdehyde (MDA) rises. Luo and colleagues found that NMN treatment decreased MDA in aged mouse liver. Additionally, the activity of the antioxidant enzyme Sod2 was increased in response to NMN treatment, suggesting an alleviation of oxidative stress.
Sirtuins are a family of enzymes that serve an array of functions associated with cell survival and, thus, are associated with increased longevity. Sirtuin 3 (Sirt3) plays a crucial role in regulating mitochondria, which provides defenses against oxidative stress. Luo and colleagues found that the effects of NMN were mediated by Sirt3, as when they genetically removed Sirt3 from mice, many of the effects of NMN were abolished. Indeed, Sirt3 uses NAD+ as fuel, and NMN is the substrate needed to synthesize and increase levels of NAD+.
NMN Enhances Adaptive Homeostasis
A protein called Nrf2 is one of the most important regulators of oxidative stress. Under oxidative stress, Nrf2 is usually transferred to the cell nucleus, where it activates antioxidant genes. By activating antioxidant genes, Nrf2 plays a protective role against oxidative stress and maintains homeostasis (physiological balance). Luo and colleagues found that Nrf2 failed to relocate to the nucleus in response to liver injury in aged mice, suggesting a lack of protective response. However, when the liver-injured aged mice were treated with NMN, Nrf2 was successfully transferred to the nucleus. These findings suggest that NMN enhances adaptations against oxidative stress and maintains homeostasis.
Can NMN Slow Liver Aging in Humans?
Previous studies have shown that NMN slows aspects of liver aging in rodents. NMN inhibits the onset of liver fibrosis (scarring) in mice by improving telomere length. In humans, NMN has been shown to reduce blood fat levels, which has been linked to fatty liver disease. However, another study showed that NMN does not affect liver fat mass. Thus, while NMN may reduce oxidative stress and prevent liver damage and fibrosis in rodents, it does not seem to affect fatty liver disease in humans. This calls for more studies examining liver function in humans in response to NMN treatment.
*Special note - This article is for informational purposes only and cannot replace a doctor's treatment diagnosis and advice. It should not be regarded as a recommendation or proof of efficacy of the medical products involved. If it involves disease diagnosis, treatment, and rehabilitation, please be sure to go to a professional medical institution to seek professional advice.
Previous: NMN Demonstrates Potential Efficacy in a Glaucoma Model
Next: Study Finds NMN Protects Intestinal Wall from Cancer Radiation Therapy Injury
by GSHWORLD
GSH Bio Tech is China Biological API Manufacturer. China NMN powder suppliers & best NMN raw material Factory.













