NMN Reduces Metabolic Impairment in Male Mouse Offspring from Obese Mothers
Highlights
Impaired glucose tolerance due to maternal and post-weaning high-fat diet was significantly improved by only 8 days of NMN treatment in mice.
In offspring of obese mouse mothers, liver lipid accumulation was reduced due to NMN treatment in regular and high-fat diet-fed offspring.
The benefits of NMN appear to have been partly mediated by the modulation of genes with roles in fat transport and metabolism in the liver.
NMN could be a therapeutic approach for treating metabolic disease due to maternal and post-weaning over-nutrition, even in late adulthood.
Extensive evidence links increased maternal obesity to adverse offspring metabolic and cardiovascular outcomes, with increased risk for obesity and metabolic disease like non-alcoholic fatty liver disease. To treat such metabolic impairments, there has been recent interest in using boosting agents like Nicotinamide Mononucleotide (NMN) for the vital molecule nicotinamide adenine dinucleotide (NAD+). Previous research shows that, in female offspring of obese mouse mothers, NMN injection improved metabolism and reduced fat accumulation. However, no studies to date have specifically examined the effects of NMN on metabolism in male offspring from obese mouse mothers.
Uddin and colleagues published an article in the journal Cells showing that only three weeks of NMN administration was able to reduce fat mass, liver and plasma levels of a type of fat called triglyceride, and to improve glucose tolerance in mice with severe metabolic impacts due to the combination of maternal obesity and chronic high fat diet consumption. The research team based out of The University of New South Wales in Sydney, Australia, showed that the benefits of NMN appear to have been partly mediated by the modulation of genes with roles in fat transport and metabolism in the liver. “These data support the use of NMN as a potential method to combat the intergenerational consequences of the obesity epidemic,” said the authors.
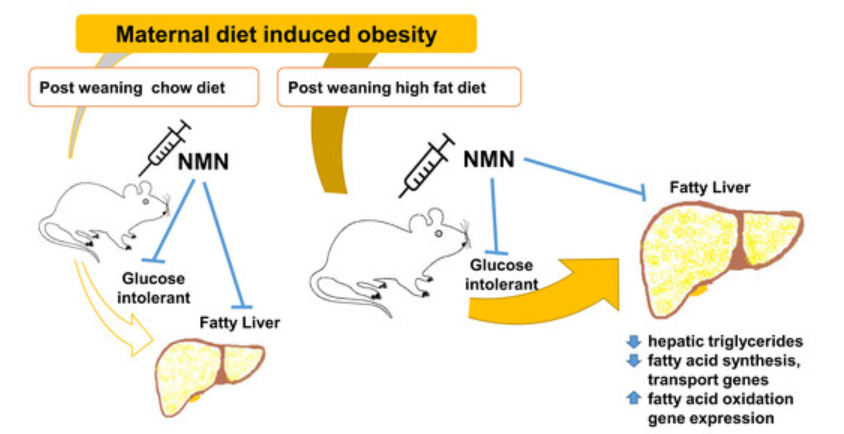
Obesity can be passed onto offspring
Parental obesity is considered a key contributor to the risk of being overweight or obese in early life. Also, it has been reported that early life developmental issues strongly associate with the prevalence of obesity and related metabolic disorders later in life. Furthermore, these impacts are known to be exacerbated by post-weaning high-fat diet consumption. While these long-term impacts of maternal obesity and chronic post-weaning high-fat diets for offspring underline the crucial need for intervention, the appropriate intervention for an individual will likely depend on many variables such as gender, age, and severity of disease features.
Treating metabolic disease with NAD+ precursors
Recently, it was shown that long term administration of NMN in drinking water successfully reversion age-associated body weight gain and improved energy metabolism, and insulin sensitivity. Also, research has shown that only 18 days of NMN injection improved glucose tolerance, reduced fat accumulation, and increased gene activity levels responsible for the metabolism of fat in female offspring of obese mothers.
However, no studies to date have examined the effects of NMN in obese male offspring. Investigating both male and female offspring is important due to the differences that have been reported in both human and rodent studies on the scale of changes in growth and adiposity of offspring. After observing improvements in metabolic performance in female offspring, Uddin and colleagues were interested in investigating whether short term NMN administration has any effect on male offspring even after long-term post-weaning exposure to a high-fat diet that caused extreme obesity.
NMN cancels out maternal and post-weaning diet effects
So, the Australian research team examined whether, under these more chronic conditions, short-term NMN (500 mg/kg body weight) administration could exert beneficial effects in male offspring consuming a high-fat diet. In this study, mice exposed to both maternal obesity and chronic post-weaning high fat diet were twice as heavy as mice fed normal diets. The male offspring of obese mothers were significantly heavier than those from lean mothers regardless of their post-weaning diet, indicating their having an obese mother facilitated weight increase.
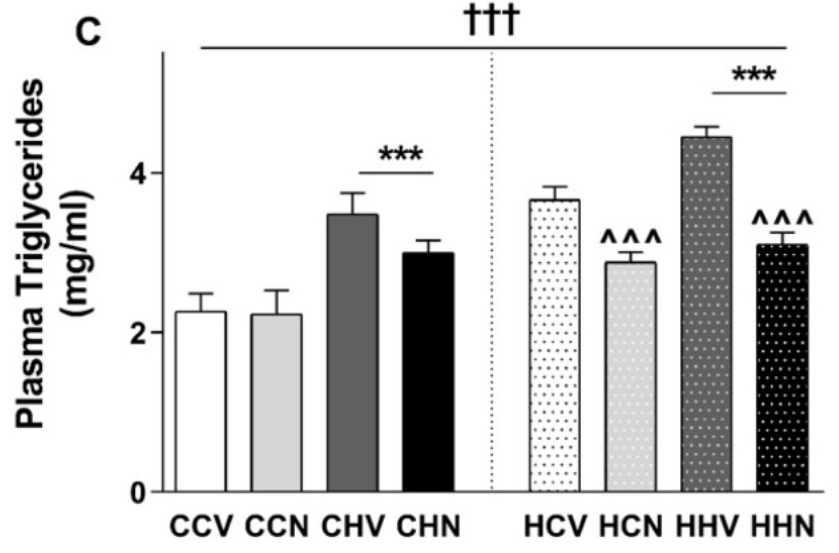
Also, significant maternal diet effects were observed in the liver, quadriceps muscle, and retroperitoneal fat. Uddin and colleagues found that three weeks of NMN administration was able to reduce fat mass. Also, NMN mitigated high liver and plasma levels of fat compounds called triglycerides that lead to cardiovascular complications in mice with severe metabolic impacts due to the combination of maternal obesity and chronic high fat diet consumption.
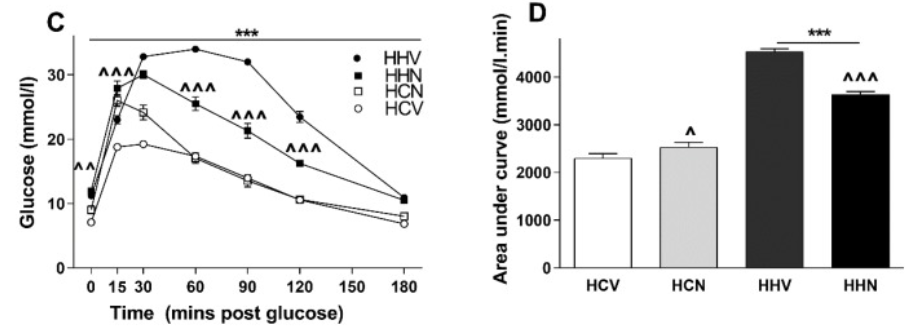
The researchers also examined the effects of short term NMN treatment (eight days). The effect of NMN on glucose intolerance, an indicator of prediabetes where the body doesn’t dispose of blood sugars, appeared to vary with the maternal dietary status. In the offspring of obese mothers, the increased glucose intolerance in mice consuming a high-fat diet post-weaning was significantly improved in the group receiving NMN injection. On the other hand, in offspring of obese mothers who consumed chow post-weaning, there was a significant increase in blood glucose concentrations, albeit briefly, in the mice receiving NMN, indicative of glucose intolerance. Overall, it appears that NMN was beneficial in those that were most metabolically compromised.
“Overall, our data clearly highlight the negative effects of maternal and long term post-weaning high-fat diet on male offspring, and point to beneficial metabolic outcomes induced by a short period of NMN administration,” said the authors. “Our data invite further work on the potential for mitochondrial activators as later-life interventions in conditions of maternal obesity and related complications such as diabetes and fatty liver disease.
*Special note - This article is for informational purposes only and cannot replace a doctor's treatment diagnosis and advice. It should not be regarded as a recommendation or proof of efficacy of the medical products involved. If it involves disease diagnosis, treatment, and rehabilitation, please be sure to go to a professional medical institution to seek professional advice.
Previous: NAD+ Precursor Improves Energy Production in Muscle of Diabetes Patients
Next: Enhancing NAD+ Can Restore Age-Related Muscle Degeneration in Human Cells
by GSHWORLD
GSH Bio Tech is China Biological API Manufacturer. China NMN powder suppliers & best NMN raw material Factory.













