NAD+ Precursor Improves Energy Production in Muscle of Diabetes Patients
Type 2 diabetes patients can have many complications, one of them often being weak muscles. This can be attributed in part to shotty mitochondria, the power generators of the cell. These cellular structures drive metabolism, a process dependent on the critical, energetic compound nicotinamide adenine dinucleotide (NAD+). The life-sustaining activities mediated by NAD+ can be boosted with NAD+ precursors, at least in animals. But whether NAD+ precursors can refuel NAD+ levels to recover mitochondrial function can translate into humans let alone diabetes patients hasn’t really been tested.
Research by van de Weijer and colleagues from Maastricht University Medical Center of the Netherlands published in the journal Diabetes testing the NAD+ precursor acipimox in type 2 diabetes patients. This research team showed for the first time that acipimox can convalesce mitochondria in the skeletal muscle of type 2 diabetes patients.
“To the best of our knowledge, this study is the first to demonstrate that NAD+ boosters can also directly affect skeletal muscle mitochondrial function in humans,” said the researchers in their study. These results suggest a strong potential for novel, safe NAD+ precursors to boost mitochondrial function and facilitate healthy aging in humans.
Clinically approved NAD+ precursor may mitigate mitochondrial health
Acipimox is a nicotinic acid analo used to treat hyperlipidemia, a condition in which there are high levels of fat particles (lipids) in the blood. However, besides the known effects of acipimox on the breakdown of fat particles, acipimox is also an NAD+ precursor. Since acipimox is already available for clinical use in humans, van de Weijer and colleagues tested whether it improves mitochondrial function in humans by boosting levels of NAD+.
Acipimox re-energizes muscle mitochondria in diabetes patients
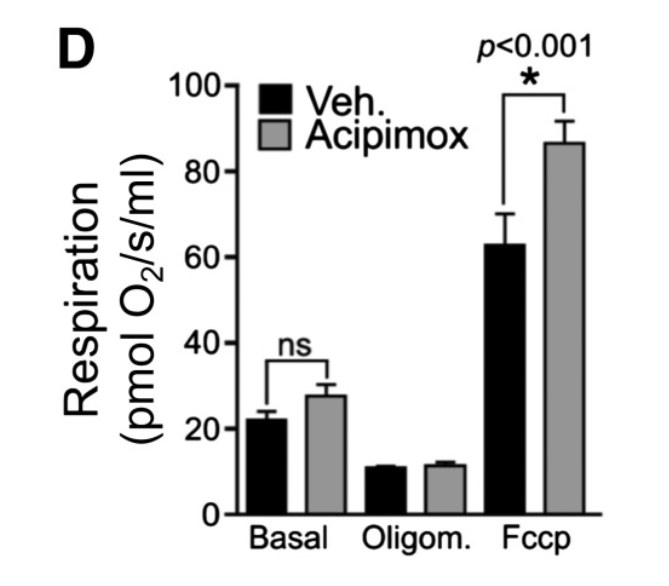
The researchers examined the effects of acipimox on mitochondrial metabolism. Overall, they saw improvements in metabolism in muscle biopsies from these patients compared to those that went untreated. They also had increased levels of ATP, a molecule that transports the energy necessary for all cellular metabolic activities.
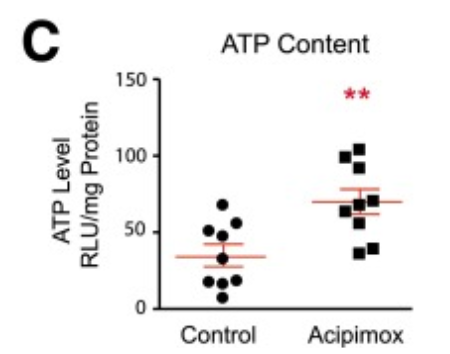
In addition, acipimox had beneficial effects on mitochondrial function in these patients. This was accompanied by activation of mitochondrial genes that overlap with the gene sets induced by NAD+ boosters, such as nicotinamide riboside (NR) and Nicotinamide Mononucleotide (NMN), in animal studies. They also saw an enrichment in the activation of genes that drive energy metabolism in muscle biopsies of acipimox-treated patients. The researchers replicated many of these results with mouse muscle cells cultured in a dish with acipimox for 24 hours.
In addition, a signaling pathway that repairs and improves mitochondrial function and metabolic health was activated in both human muscle biopsy specimens and cultured mouse muscle cells treated with acipimox. Activation of this process, called the mitochondrial unfolded protein response, by NAD+ precursors had previously been shown in animals but never in humans. “To the best of our knowledge, this study is the first to demonstrate that as in animals treated with NAD+ boosters, the mitochondrial unfolded protein response is present in humans,” said the researchers in the article.
What's next for NAD+ precursor treatment in humans?
These results suggest a strong potential for novel, safe NAD+ precursors to enhance mitochondrial function in humans. Although acipimox itself may not be a suitable candidate, new NAD+ precursors, such as NR and NMN, devoid of such side effects like skin flushing — feelings of warmth and rapid reddening — may potentially act as mitochondrial boosters.
Human clinical trials with such agents are urgently needed given that interventions targeting mitochondrial function, such as caloric restriction, exercise, and resveratrol, have been shown to be effective in improving metabolic health. This work shows that NAD+ precursors should be tested for their ability to boost mitochondrial function and improve metabolic health in human subjects, such as type 2 diabetes patients.
*Special note - This article is for informational purposes only and cannot replace a doctor's treatment diagnosis and advice. It should not be regarded as a recommendation or proof of efficacy of the medical products involved. If it involves disease diagnosis, treatment, and rehabilitation, please be sure to go to a professional medical institution to seek professional advice.
Previous: A Small Molecule That Activates NAMPT Boosts NAD+ Levels
Next: NMN Reduces Metabolic Impairment in Male Mouse Offspring from Obese Mothers
by GSHWORLD
GSH Bio Tech is China Biological API Manufacturer. China NAD+ powder suppliers & best NAD+ raw material Factory.














