Glutathione and tumors
Glutathione (GSH) is a tripeptide containing γ-amide bonds and sulfhydryl groups, composed of glutamic acid, cysteine and glycine, and exists in almost every cell of the body.
Glutathione and tumors, the direct cause of tumor death is not due to tumors, many reasons are due to malnutrition. Glutathione not only emphasizes the supplement of macronutrients but also is a good micronutrient supplement, and can achieve the effect of anti-oxidation (ROS).
In a double-blind controlled trial at the National Cancer Institute of Thailand, a total of 42 cancer patients (41-63 years old) receiving intravenous chemotherapy were randomized. Patients received 40 g of WPI per day plus zinc and selenium (intervention group, n = 23) or maltodextrin oral snack (control group, n = 19) for 12 weeks.
Nutritional status, glutathione levels, immune and inflammatory markers were assessed at baseline at 6 and 12 weeks. The whey protein supplementation group significantly increased albumin (2.9%) and immunoglobulin G (4.8%) levels at week 12 compared to the control group.
The percent change in glutathione levels was significantly lower in the control group (6.0%), whereas the intervention group had a significant time-dependent increase (11.7%).
Whey protein supplementation improved nutritional status scores in the intervention group compared with the control group.
These data suggest that whey protein supplementation can increase glutathione levels and improve nutritional status and immune function in cancer patients undergoing chemotherapy.
These results will facilitate malnutrition risk prevention and improve protein status, including immune function, during chemotherapy.
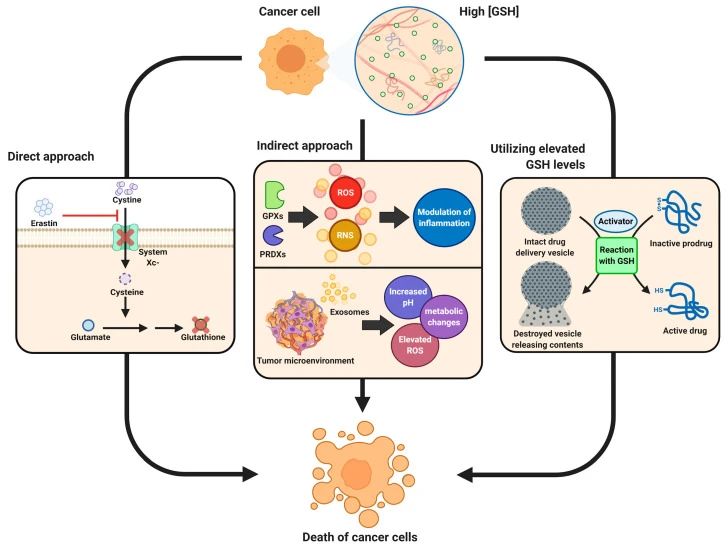
Direct therapy aims to block GSH synthesis leading to cellular ferroptosis or more effective chemotherapy;
indirect approaches target the inflammatory response or tumor microenvironment to make the tumor more susceptible to immune system responses and immunotherapy;
high levels of GSH and Possibly other active molecules in tumor cells are used in drug-directed approaches, in which drugs are activated only after they enter the activation response of tumor cells using GSH, or are neutralized after being activated by other factors.
Recent studies have highlighted the important role of GSH in key signal transduction responses such as cell differentiation, proliferation, apoptosis, ferroptosis and immune function.
Molecular changes in the glutathione antioxidant system and disturbances in glutathione homeostasis are associated with tumor initiation, progression, and treatment response.
During complex and tightly controlled oxidative processes, such as glycolysis in the cytoplasm, citric acid cycle and oxidative phosphorylation in the mitochondria, electrons are used to power the synthesis of ATP, which then supports the high energy demands of tumor cells.
However, during the oxidation of nutrients, electrons can escape and interact with oxygen to form ROS.
In most cell types, including tumor cells, mitochondrial redox reactions are the main source of ROS. ROS are produced by NADPH oxidase (NOX) enzymes, located on the plasma membrane of cells, mainly in neutrophils and vascular endothelial cells.
To avoid the toxic effects of ROS on DNA, proteins, and lipids, cells have evolved a wide range of antioxidant mechanisms, especially in tumor cells with high redox activity.
Many ROS defense mechanisms rely heavily on GSH. The burden of reactive nitrogen species (RNS) in tumors will increase simultaneously with ROS.
These reactive nitrogen species are enzymatically generated by nitric oxide synthases (NOS) and distributed in tumor cells, stromal cells or infiltrating cells. in immune cells.
NOS activity produces NO gas, which undergoes non-enzymatic reactions with glutathione, major non-protein thiols, and cysteine residues in protein thiols to form s-nitrosoglutathione (GSNO) and s-nitroso Nitrothiols, this process is called S-nitrosylation.
Through these responses and its interactions with many proteins, glutathione is one of the master regulators of cancer cell development, progression, response to therapy and its environment, leading to many therapeutically targetable pathways.
This also provides good theoretical support for many glutathione-based dietary nutrition treatments and supplement treatments.
*Special note - This article is for informational purposes only and cannot replace a doctor's treatment diagnosis and advice. It should not be regarded as a recommendation or proof of efficacy of the medical products involved. If it involves disease diagnosis, treatment, and rehabilitation, please be sure to go to a professional medical institution to seek professional advice.
by GSHWORLD
GSH Bio Tech is China Biological API Manufacturer. China Glutathione powder suppliers & best Glutathione raw material Factory.













